Description
Dosage form
Ophthalmic Solution (Eye Drops)
Category
Antibiotic + Steroid
Mofo DX
0. 1% w/v Moxifloxacin is a fourth generation fluoroquinolone controls
infection by inhibitting the dna gyrase & topoisomerase iv.
Dexamethasone a potent corticosteroid effectively controls the
inflammation by inhibiting the release inflammatory mediators.
Indications
Mofo DX is indicated for steroid-responsive inflammatory ocular
conditions for which a corticosteroid is indicated and where bacterial
infection or a risk of bacterial ocular infection exists. The combination can
also be used for post-operative inflammation and any other ocular
inflammation associated with infection.
Dosage
One or two drops instilled into the conjunctival sac(s), every 4 to 6 hours.
during the initial 24 to 48 hours. The dosage may be increased to I or 2
drops every two hours. Frequency must be decreased gradually or
warranted by improvement in clinical signs. Care should be taken not to
discontinue the therapy prematurely.
Contraindications
Mofo DX is contraindicated in epithelial herpes simplex keratitis
(Dendritic keratitis), vaccinia, varicella, and in many other viral diseases
of the conjunctiva and cornea, Mycobacterial infection of the eye and
fungal diseases of ocular structures and in individuals hypersensitive to
any of the components of the medication.
Warnings & precautions
Prolonged use of steroids may result in glaucoma with damage to the
optic nerve, defects in visual acuity and fields of vision, and posterior
subcapsular cataract formation.
Adverse effects
The most frequently reported drug-related undesirable effects seen with
moxifloxacin are conjunctival irritation, increased lacrimation, keratitis
and papillary conjunctivitis.
is a combination of Moxifloxacin 0.5% w/v and Dexamethasone
(Moxifloxacin with Dexamethasone Eye Drops)
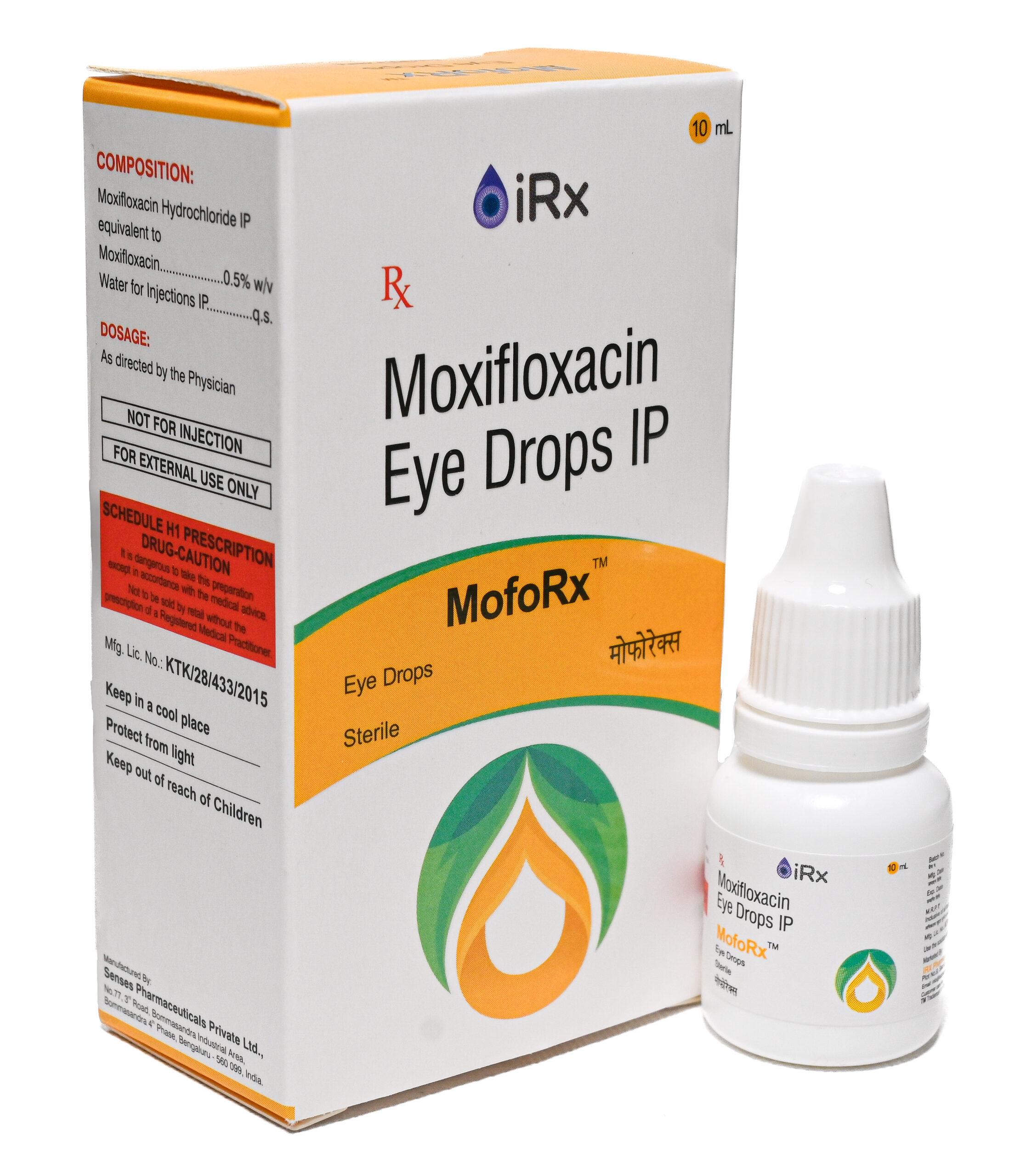
Moxifloxacin Hydrochloride IP equivalent to Moxifloxacin……………….. 0.5%w/v
Dexamethasone Sodium Phosphate IP equivalent to
Dexamethasone Phosphate…………………………………………………..….0.1%w/v
Water for Injections IP……………………………………………………………………..…. q.s
Eye Drops
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory
COMPOSITION:
Mofo DX
Storage
0 Store below 30 C, Do not freeze, Protect from light
DO NOT FREEZE
KEEP OUT OF REACH OF CHILDREN
NOT FOR INJECTION
FOR EXTERNAL USE ONLY
Solution to be used with in one month after opening the container.
Presentation
Mofo DX eye drops is available in sterile opaque plastic bottle with a cap
containing 5 mL of the solution
Directions for use :
Remove the cap, dispense drops with gentle pressure.
Turn the tamper proof cap anti-clockwise to break the seal.
Replace the cap immediately after every use.
RDMPi-01
Manufactured by :
No.77, 3rd Road, Bommasandra Industrial Area
Bommasandra 4th Phase, Bengaluru – 560 099, India.
Senses Pharmaceuticals Private Ltd.,
Month of publication of pack insert: June 2024
Marketed by:
iRx Pharmaceuticals Pvt. Ltd.
Email: info@irxpharmaceuticals.com
Customer care No.: +91 88005 12273 Mon to Sat (10 am to 5 pm)
Ophthalmic Solution (Eye Drops)
Category
Antibiotic + Steroid
Mofo DX
0. 1% w/v Moxifloxacin is a fourth generation fluoroquinolone controls
infection by inhibitting the dna gyrase & topoisomerase iv.
Dexamethasone a potent corticosteroid effectively controls the
inflammation by inhibiting the release inflammatory mediators.
Indications
Mofo DX is indicated for steroid-responsive inflammatory ocular
conditions for which a corticosteroid is indicated and where bacterial
infection or a risk of bacterial ocular infection exists. The combination can
also be used for post-operative inflammation and any other ocular
inflammation associated with infection.
Dosage
One or two drops instilled into the conjunctival sac(s), every 4 to 6 hours.
during the initial 24 to 48 hours. The dosage may be increased to I or 2
drops every two hours. Frequency must be decreased gradually or
warranted by improvement in clinical signs. Care should be taken not to
discontinue the therapy prematurely.
Contraindications
Mofo DX is contraindicated in epithelial herpes simplex keratitis
(Dendritic keratitis), vaccinia, varicella, and in many other viral diseases
of the conjunctiva and cornea, Mycobacterial infection of the eye and
fungal diseases of ocular structures and in individuals hypersensitive to
any of the components of the medication.
Warnings & precautions
Prolonged use of steroids may result in glaucoma with damage to the
optic nerve, defects in visual acuity and fields of vision, and posterior
subcapsular cataract formation.
Adverse effects
The most frequently reported drug-related undesirable effects seen with
moxifloxacin are conjunctival irritation, increased lacrimation, keratitis
and papillary conjunctivitis.
is a combination of Moxifloxacin 0.5% w/v and Dexamethasone
(Moxifloxacin with Dexamethasone Eye Drops)
Moxifloxacin Hydrochloride IP equivalent to Moxifloxacin……………….. 0.5%w/v
Dexamethasone Sodium Phosphate IP equivalent to
Dexamethasone Phosphate…………………………………………………..….0.1%w/v
Water for Injections IP……………………………………………………………………..…. q.s
Eye Drops
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory
COMPOSITION:
Mofo DX
Storage
0 Store below 30 C, Do not freeze, Protect from light
DO NOT FREEZE
KEEP OUT OF REACH OF CHILDREN
NOT FOR INJECTION
FOR EXTERNAL USE ONLY
Solution to be used with in one month after opening the container.
Presentation
Mofo DX eye drops is available in sterile opaque plastic bottle with a cap
containing 5 mL of the solution
Directions for use :
Remove the cap, dispense drops with gentle pressure.
Turn the tamper proof cap anti-clockwise to break the seal.
Replace the cap immediately after every use.
RDMPi-01
Manufactured by :
No.77, 3rd Road, Bommasandra Industrial Area
Bommasandra 4th Phase, Bengaluru – 560 099, India.
Senses Pharmaceuticals Private Ltd.,
Month of publication of pack insert: June 2024
Marketed by:
iRx Pharmaceuticals Pvt. Ltd.
Email: info@irxpharmaceuticals.com
Customer care No.: +91 88005 12273 Mon to Sat (10 am to 5 pm)
Registered Trademark


Reviews
There are no reviews yet.