Pioneering the Future of Innovative Healthcare Solutions
Showing 13–24 of 49 results
-
-
-
MofoDx
COMPOSITION:
Moxifloxacin Hydrochloride IP equivalent to Moxifloxacin……………….. 0.5%w/v
Dexamethasone Sodium Phosphate IP equivalent to
Dexamethasone Phosphate…………………………………………………..….0.1%w/v
Water for Injections IP……………………………………………………………………..…. q.s -
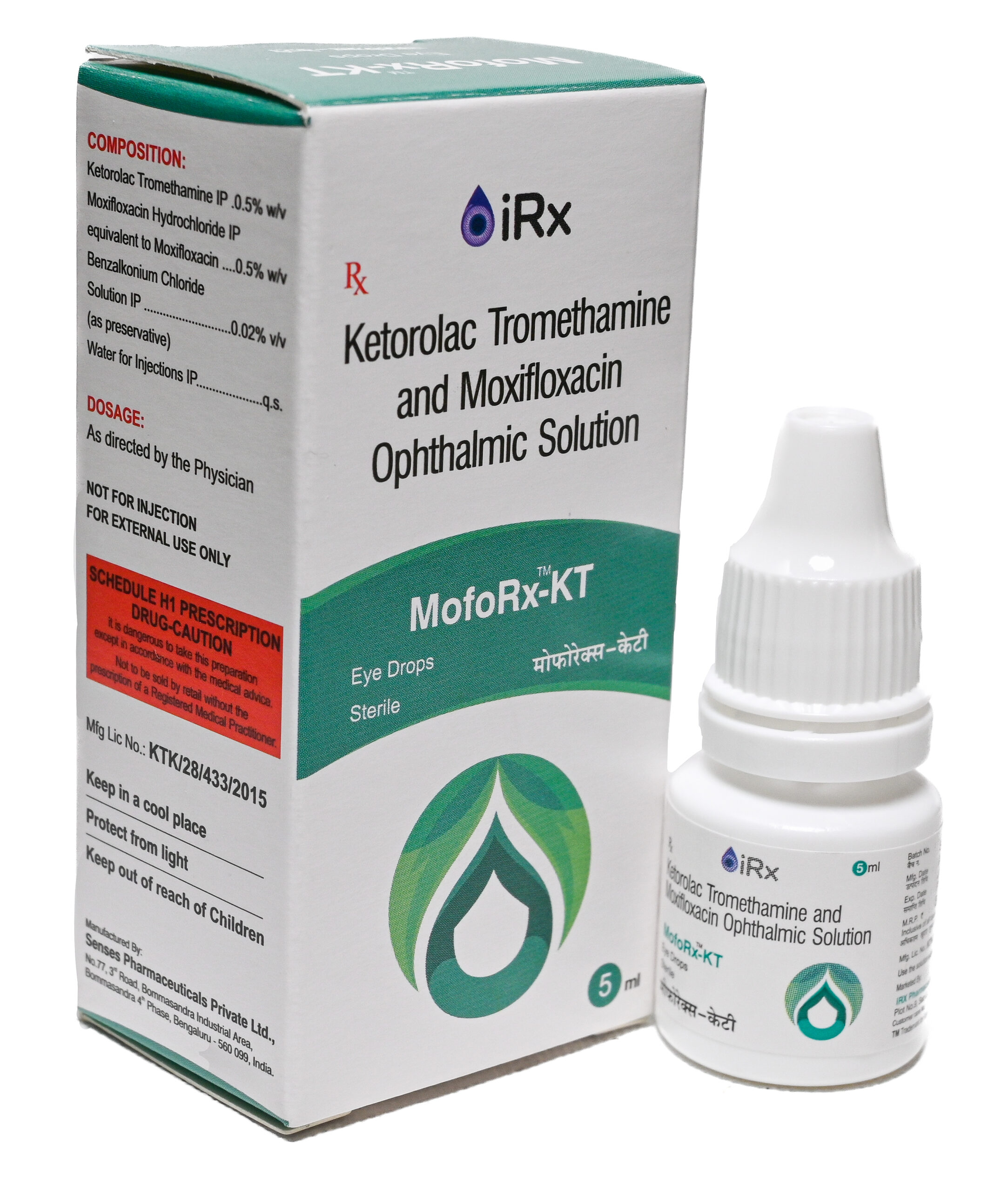
MofoRx-KT
COMPOSITION:
Ketorolac Tromethamine IP ……………………………………………………….. 0.5% w/v
Moxifloxacin Hydrochloride IP equivalent to Moxifloxacin ……………… 0.5% w/v
Benzalkonium chloride Solution IP ……………………………………………. 0.02% v/v
(As preservative)
Water for Injections IP ………………………………………………………………………. q.s. -
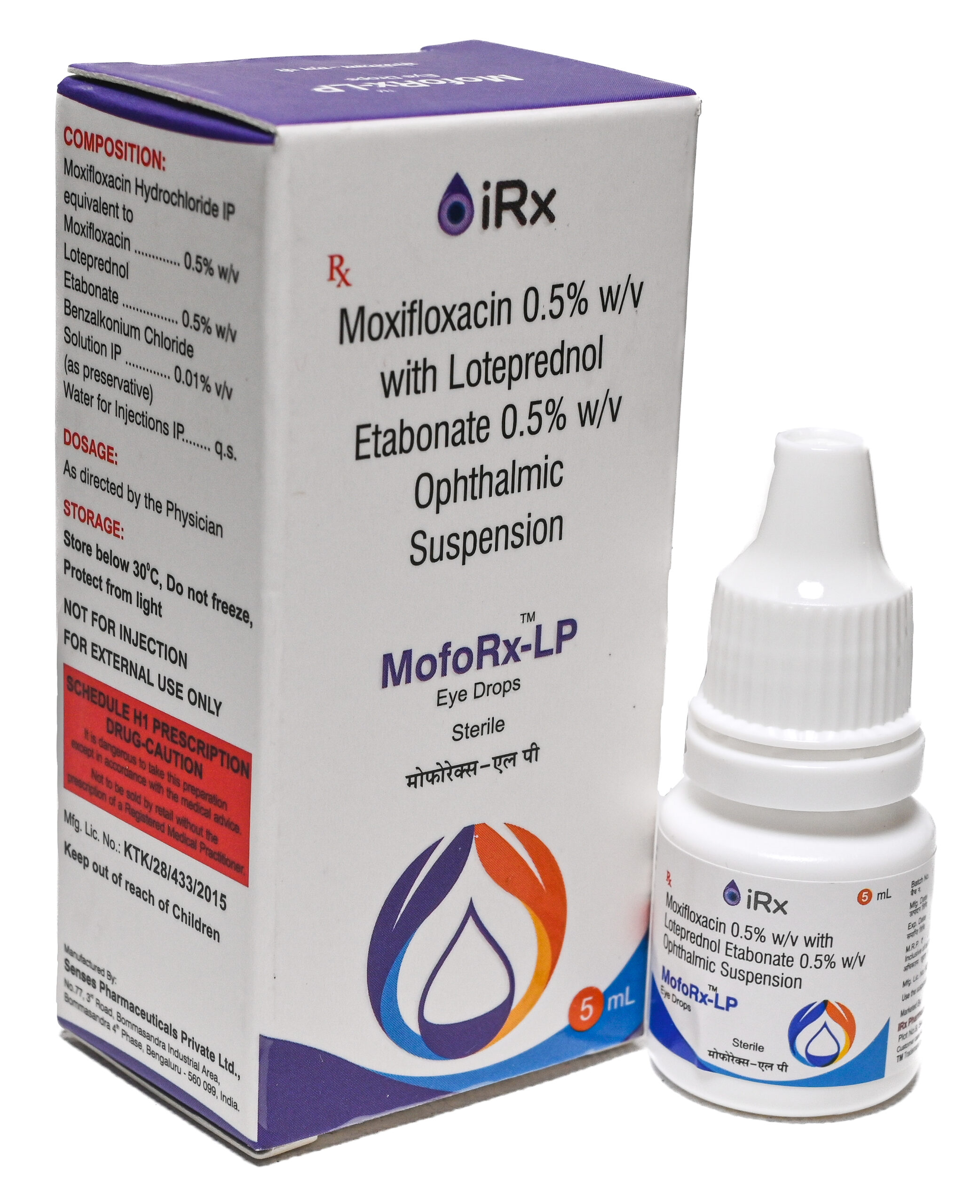
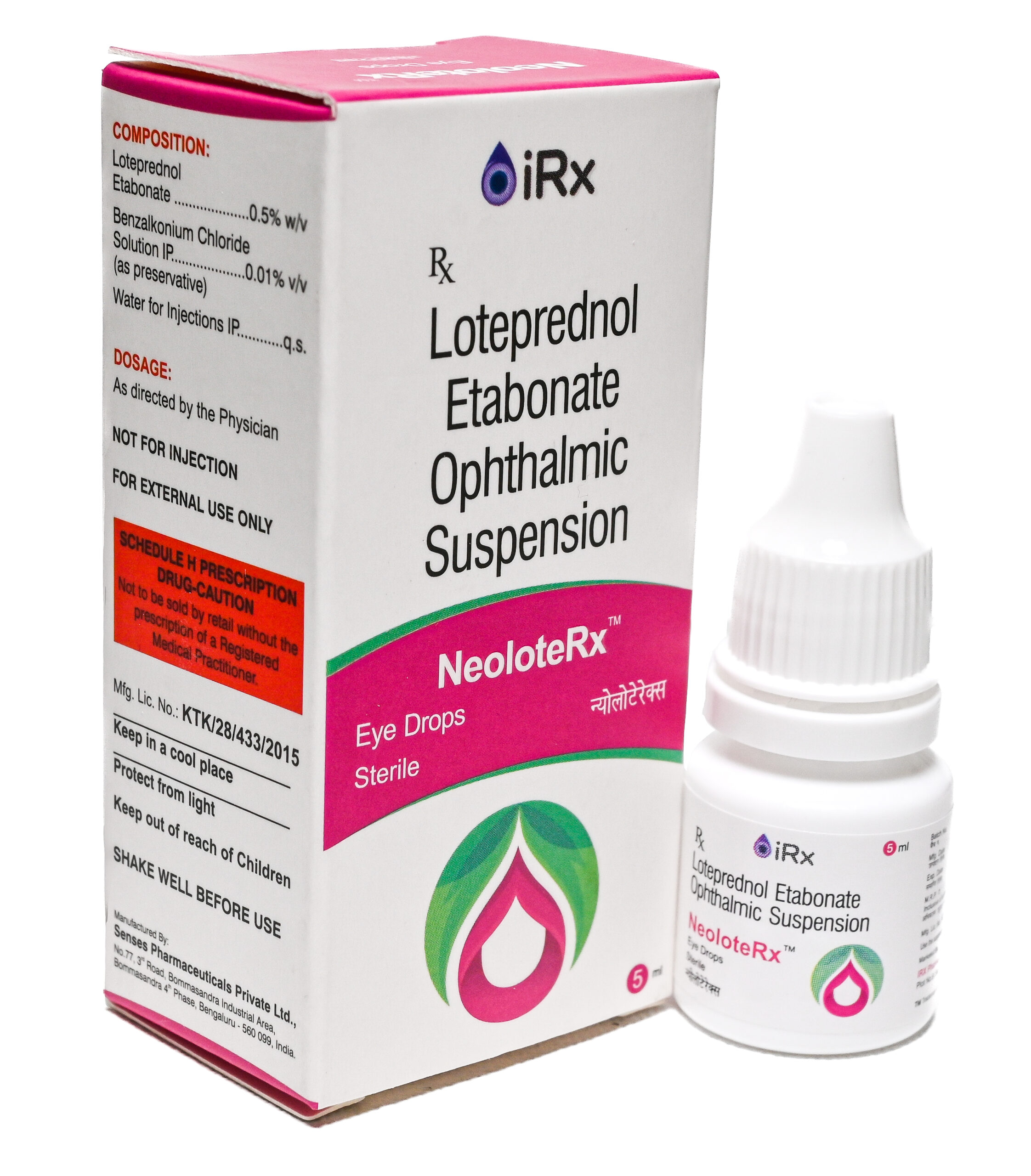
MofoRx-LP
COMPOSITION:
Moxifloxacin Hydrochloride IP Eq. to Moxifloxacin………………..…….. 0.5% w/v
Loteprednol Etabonate …………………………………………………….…….. 0.5% w/v
Benzalkonium Chloride Solution IP…………………………………………… 0.01% v/v
(As preservative)
Water for injections IP……………………………………..……………………….…… q.s. -
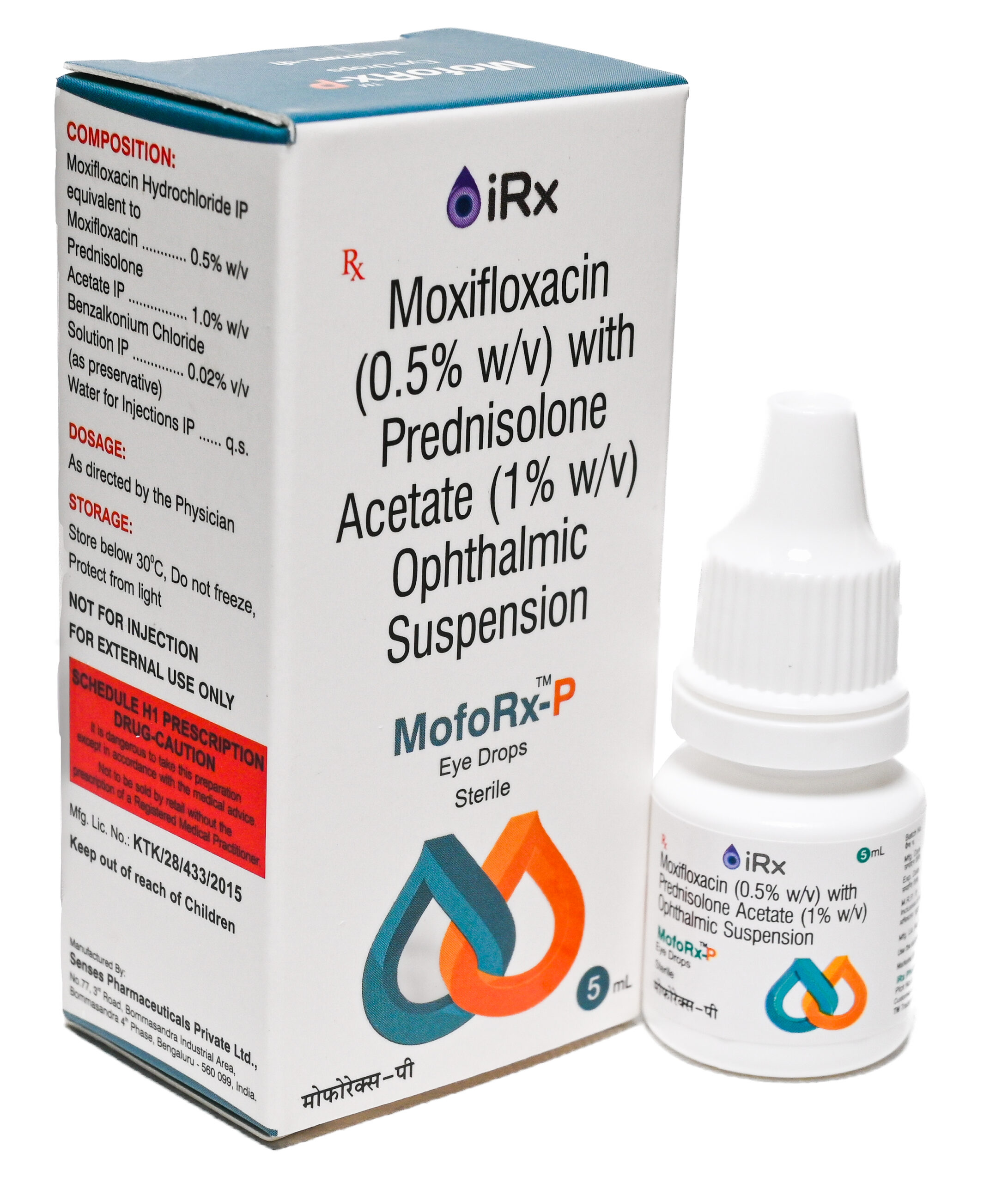
MofoRx-P
COMPOSITION:
Moxifloxacin Hydrochloride IP equivalent to Moxifloxacin ………..… 0.5% w/v
Prednisolone Acetate IP………………………………………………………. 1.0 % w/v
Benzalkonium Chloride Solution IP ………………………………………… 0.02% v/v
(as preservative)
Water for Injections IP………………………………………………………………….. q.s. -
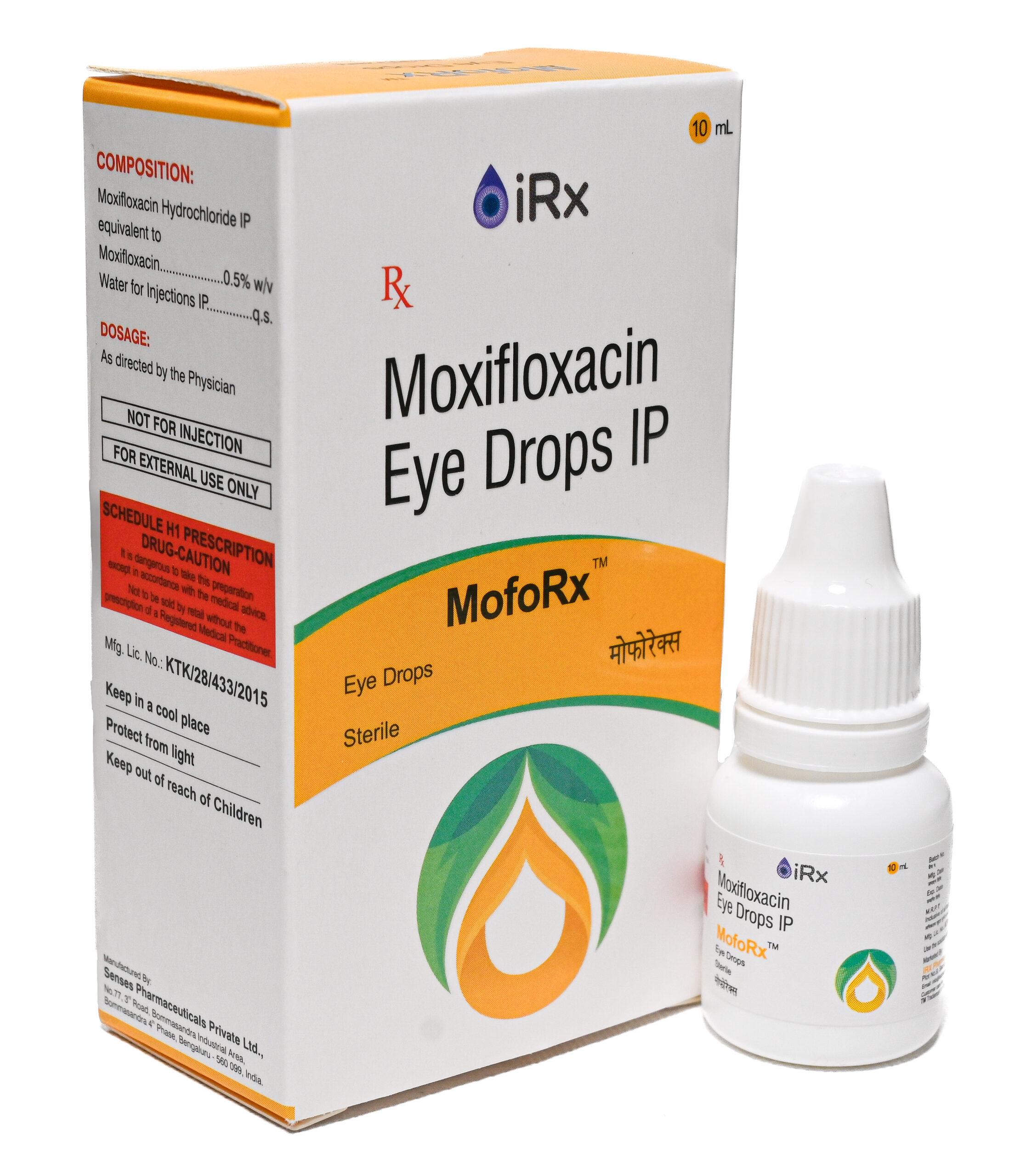
MofoRx/MofoRx-Mini
COMPOSITION:
Moxifloxacin Hydrochloride IP equivalent to
Moxifloxacin ………………………………………………………..0.5% w/v
Water for injections IP …………………………………………………… q.s. -
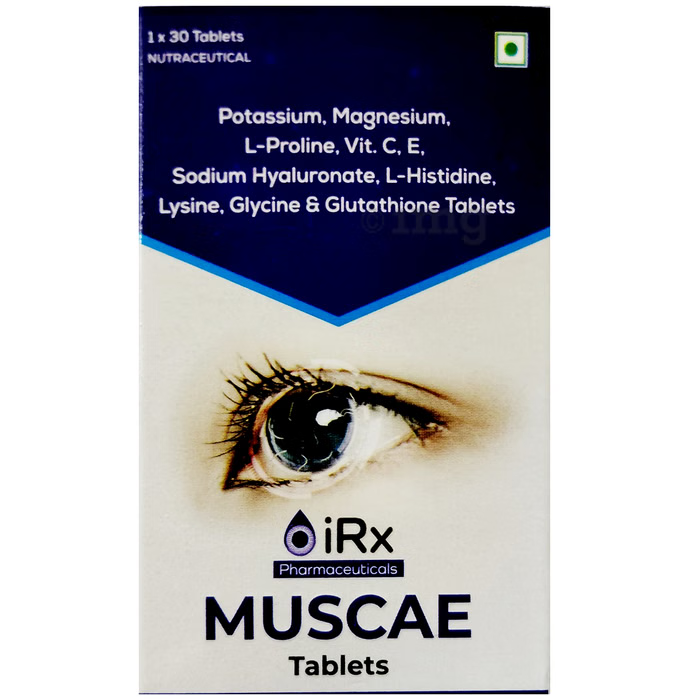
Muscae (Tablets)
COMPOSITION
Potassium (As Potassium Chloride) – 56.9mg
Magnesium (As Magnesium Sulphate) – 10mg
L Glutamic Acid – 25mg
L Ascorbic Acid (Vitamin C) – 100mg
Sodium Hyaluronate – 20mg
L-Glutathione – 10mg
L-Glycine – 10mg
L-Histine – 10mg
L-Lysine – 10mg
D-Alpha-Tocopheryl Acetate (Vitamin E) – 10mg -
-
-
-