Pioneering the Future of Innovative Healthcare Solutions
Showing 1–12 of 49 results
-
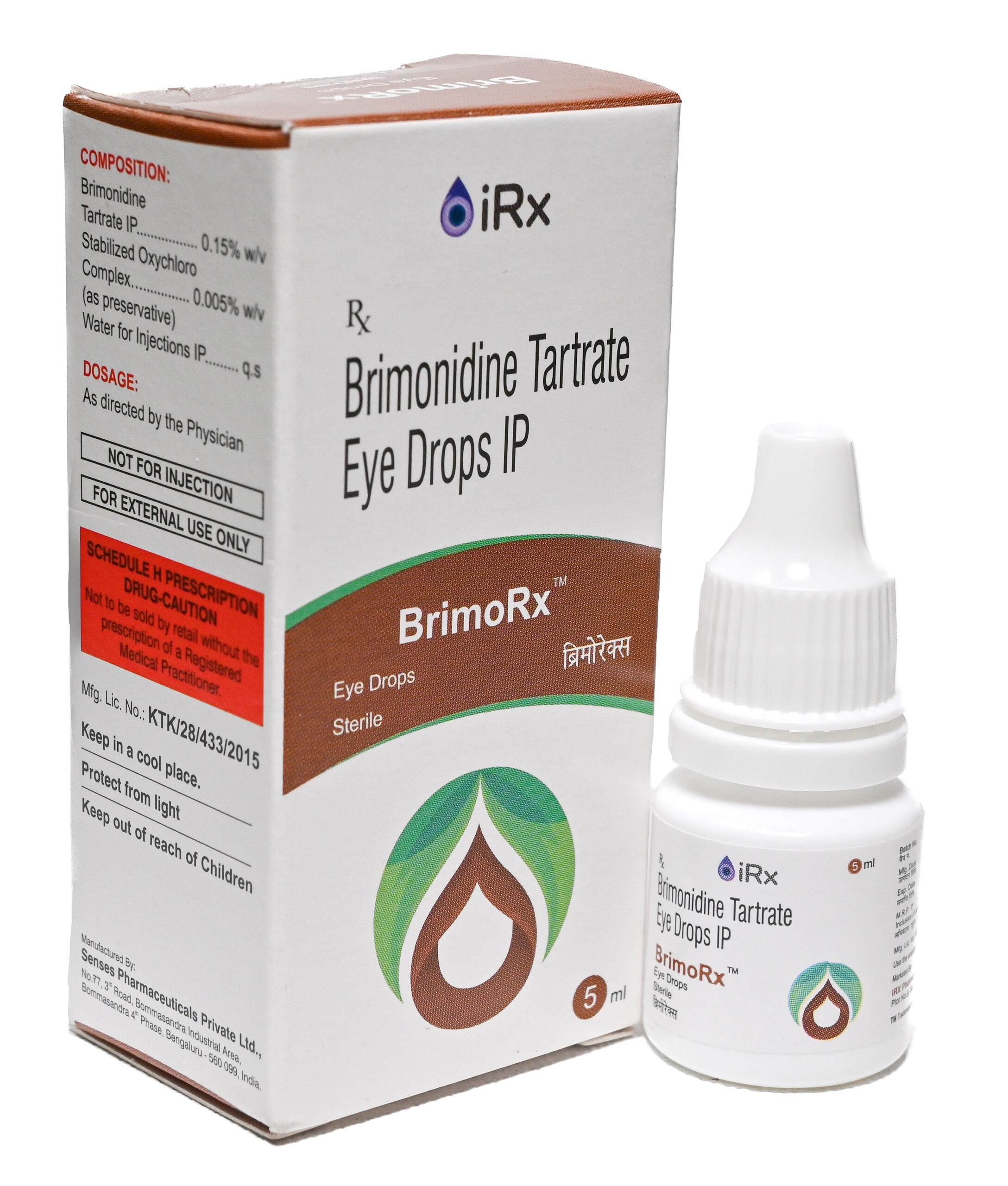
BrimoRx
COMPOSITION
Brimonidine Tartrate IP……………………………… 0.15% w/v
Stabilised Oxychloro Complex ………………… 0.005% w/v
(as preservative)
Water for Injections IP …………………………………………. q.s. -
-
ComfiRx OD
COMPOSITION
Olopatadine Hydrochloride IP Equivalent to Olopatadine ………….. 0.2 % w/v
Benzalkonium Chloride Solution IP ………………………………………… 0.02 % v/v
(As Preservative)
Water for Injections IP …………………………………………………………………..… q.s -
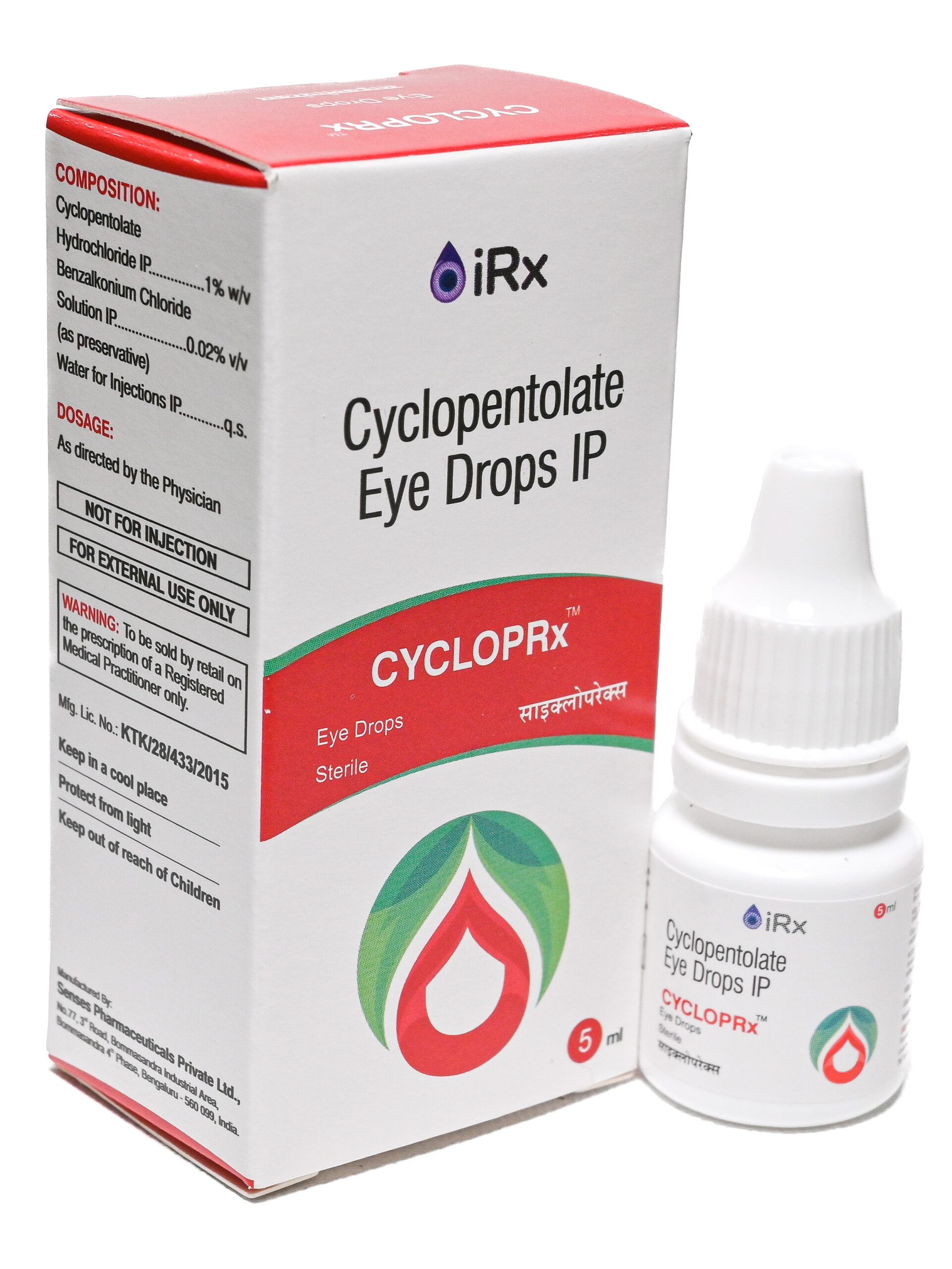
CYCLOPrx
COMPOSITION:
Cyclopentolate Hydrochloride IP ………………………………………………… 1% w/v
Benzalkonium Chloride Solution IP …………………………………………. 0.02% v/v
(as preservative)
Water for Injections IP …………………………………………………………………….. q.s. -
DorzoRx
Composition
Dorzolamide Hydrochloride IP Equivalent to Dorzolamide…. 2% w/v
Benzalkonium Chloride solution IP….. 0.02% v/v
(As Preservative)
Water for injections IP …………………………………………………. q.s. -
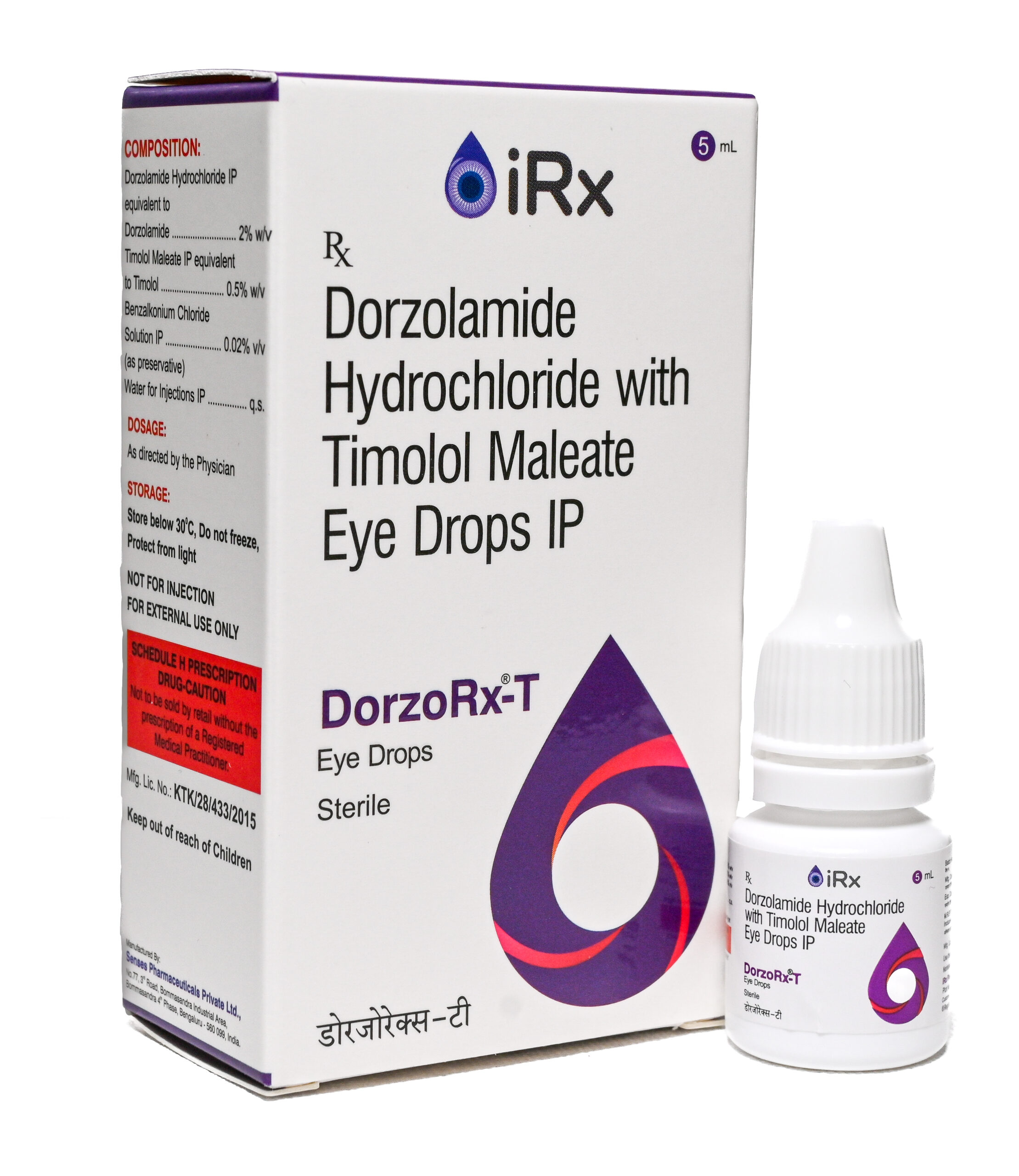
DorzoRx-T
COMPOSITION
Dorzolamide Hydrochloride IP equivalent to Dorzolamide ……………. 2% w/v
Timolol Maleate IP equivalent to Timolol ……………………………………… 0.5% w/v
Benzalkonium Chloride solution IP……………………………………………. 0.02% v/v
(as preservative)
Water for Injections IP………………………………………………………………………. q.s. -
-
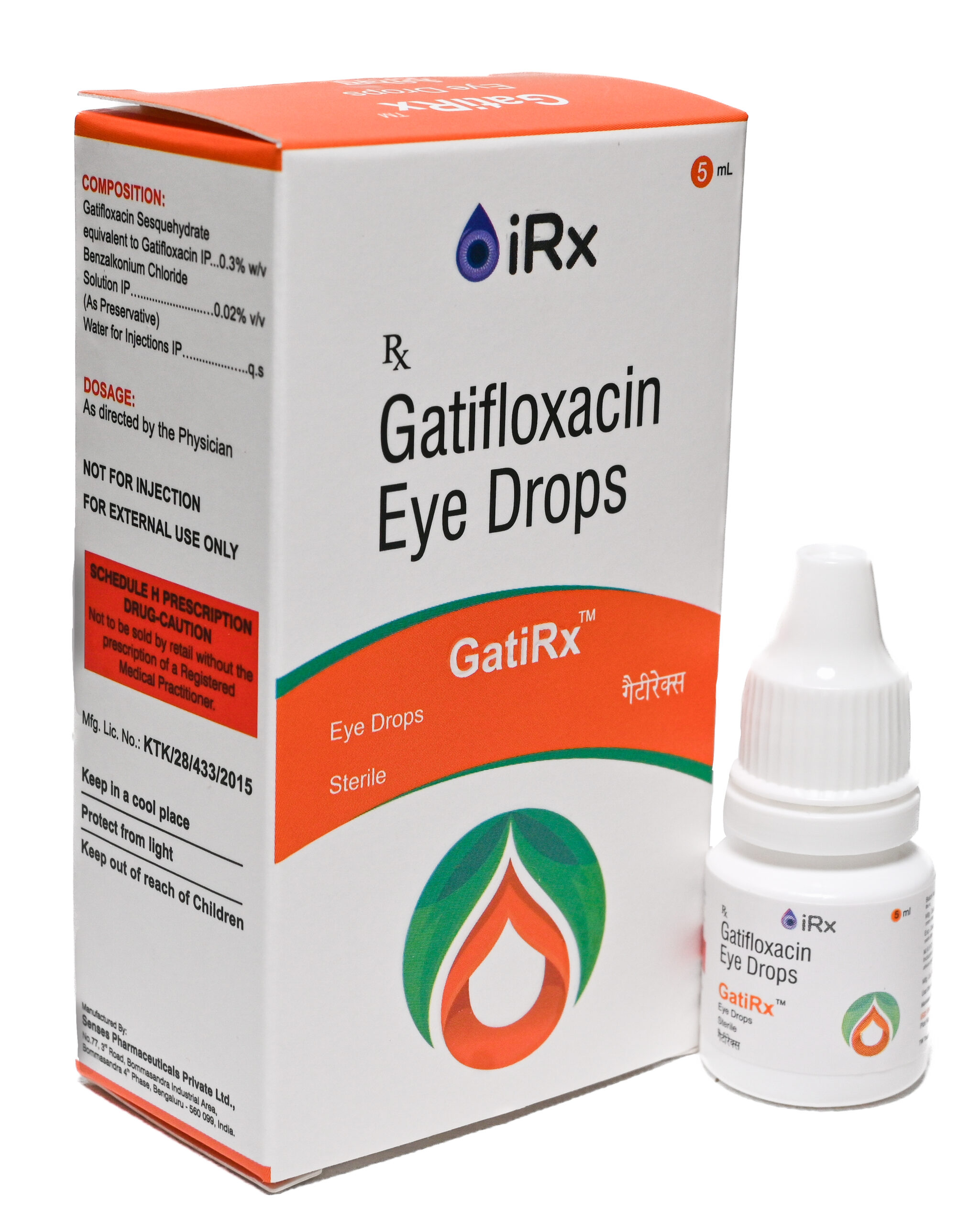
GatiRx H.S.
COMPOSITION:
Gatifloxacin Sesquehydrate eq. to Gatifloxacin ….…………………..… 0.5%w/v
Benzalkonium Chloride Solution IP …………………………………..…… 0.02% v/v
(As Preservative)
Water for Injections IP …………………………………………………………….……. q.s.
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory
(Gatifloxacin Eye Drops 0.5% w/v)
Eye Drops
GatiRx HS -
-
HyaloRx Ultra
COMPOSITION:
Sodium Hyaluronate BP ………………………………………………………………….. 0.1 w/v
Stabilized Oxychloro Complex (as preservative) …………………………… 0.005% w/v
Water for Injections IP ……………………………………………………………………….. q.s. -
HyaloRx/PF HS
COMPOSITION:
Sodium Hyaluronate BP ………………………………………………………….. 0.18% w/v
Water for Injections IP ………………………………………………………………………. q.s. -